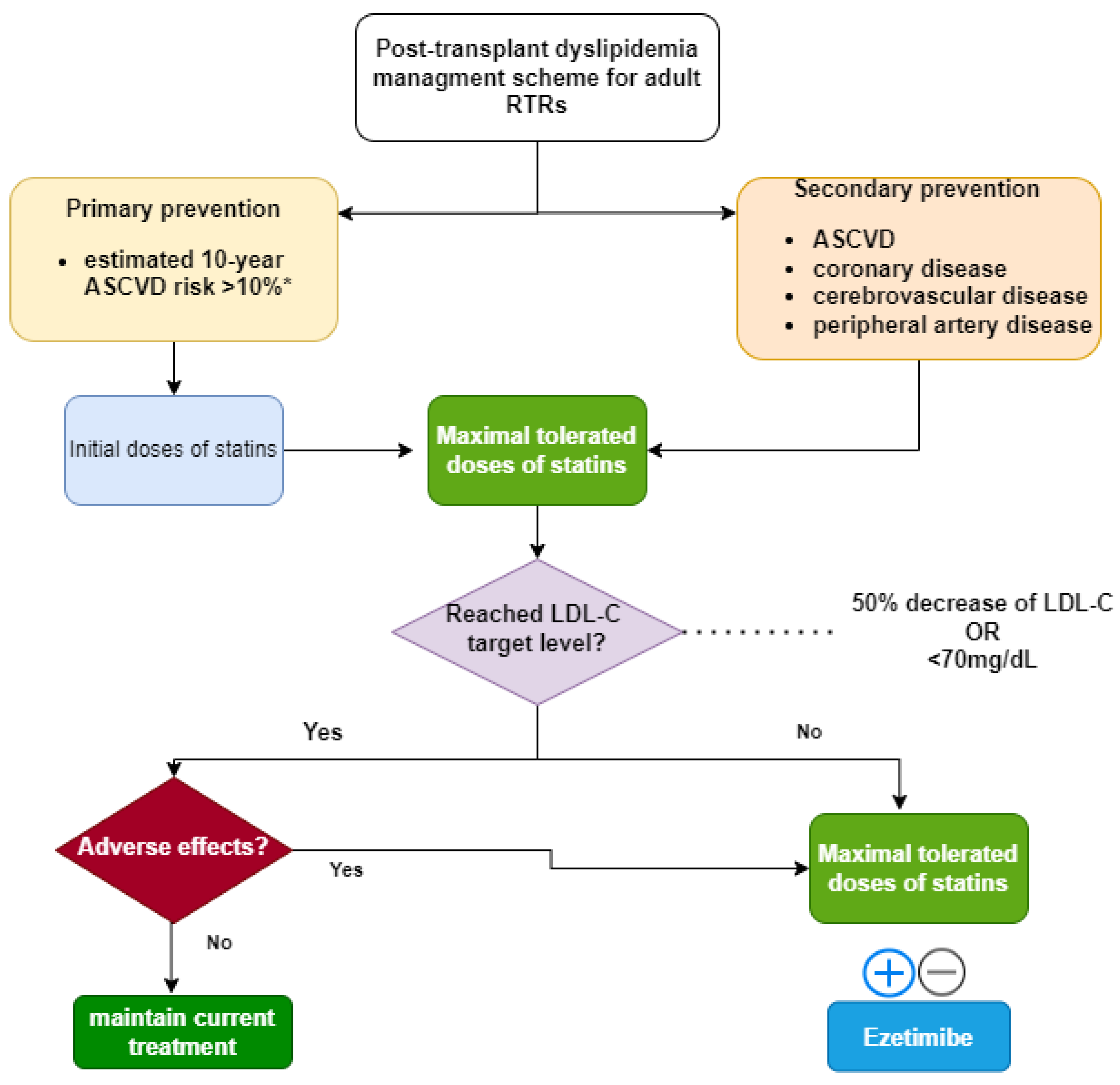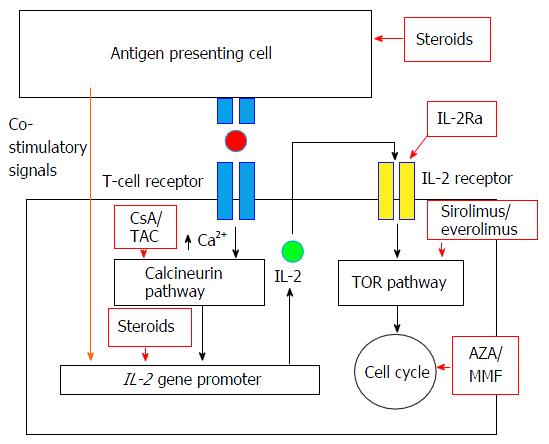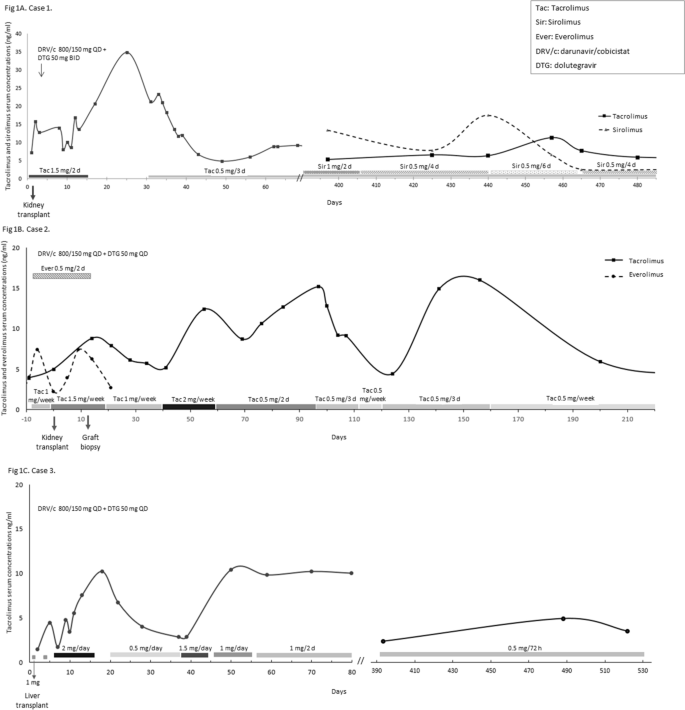
Tacrolimus, Sirolimus and Everolimus Doses in HIV-Infected Solid-Organ Recipients, Requiring a Cobicistat-Based Antiretroviral Regimen: Report of Three Cases and Review | SpringerLink

Early conversion of pediatric kidney transplant patients to everolimus with reduced tacrolimus and steroid elimination: Results of a randomized trial - American Journal of Transplantation
![PDF] Introduction of everolimus in kidney transplant recipients at a late posttransplant stage | Semantic Scholar PDF] Introduction of everolimus in kidney transplant recipients at a late posttransplant stage | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/52b7f3e549ab8e70c2079418becddcf9e9535b89/3-Table1-1.png)
PDF] Introduction of everolimus in kidney transplant recipients at a late posttransplant stage | Semantic Scholar

An open-label, randomized trial indicates that everolimus with tacrolimus or cyclosporine is comparable to standard immunosuppression in de novo kidney transplant patients - ScienceDirect
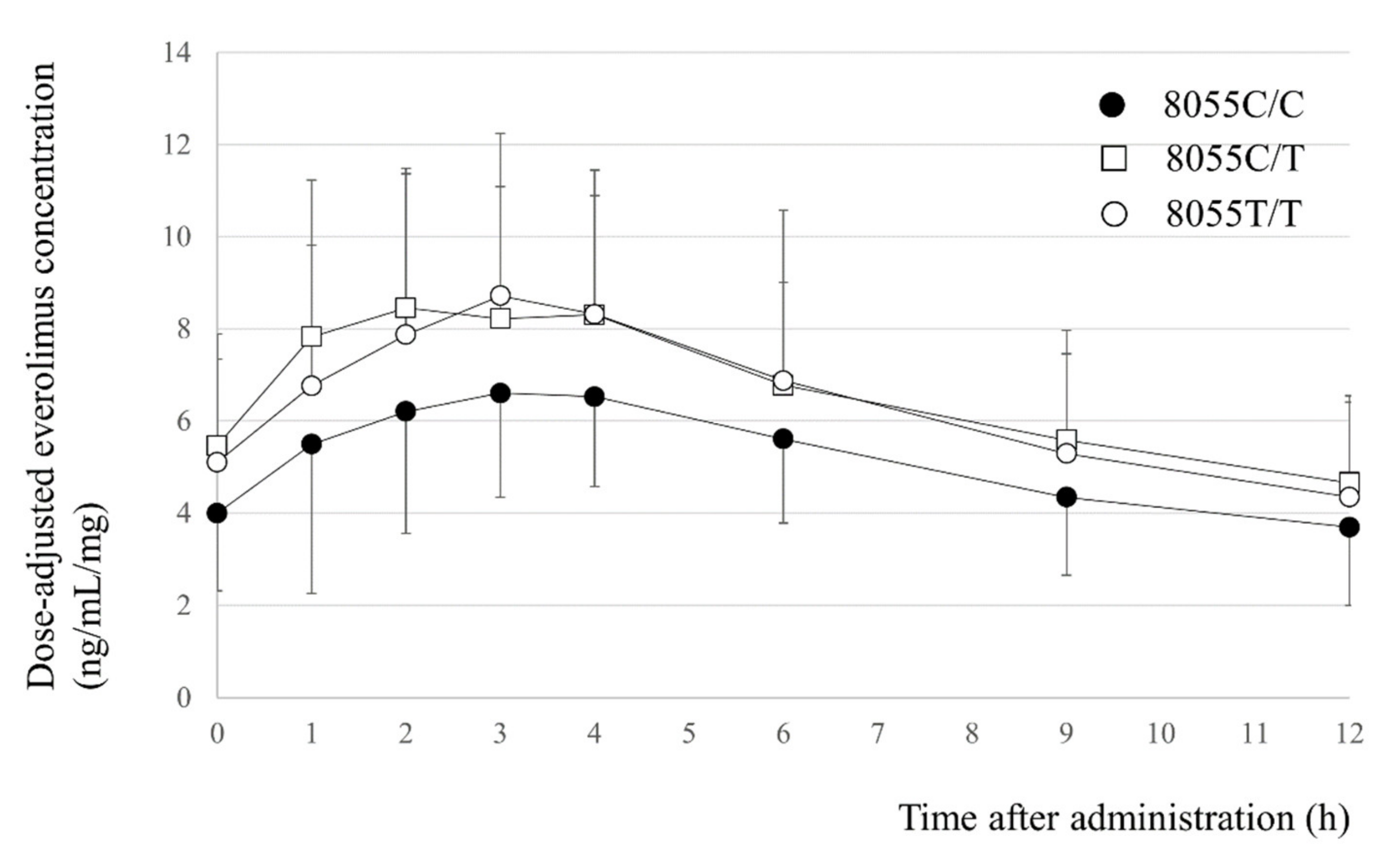
IJMS | Free Full-Text | Effects of NR1I2 and ABCB1 Genetic Polymorphisms on Everolimus Pharmacokinetics in Japanese Renal Transplant Patients

Recommendations for the use of everolimus in de novo kidney transplantation: False beliefs, myths and realities | Nefrología

Recommendations for the use of everolimus in de novo kidney transplantation: False beliefs, myths and realities | Nefrología
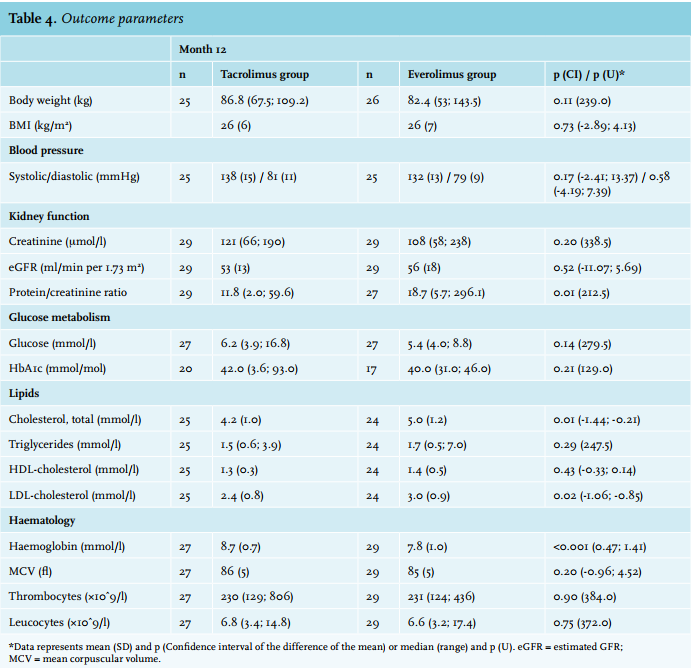
Article: Conversion from tacrolimus to everolimus with complete and early glucocorticoid withdrawal after kidney transplantation: a randomised trial (full text) - January 2018 - NJM
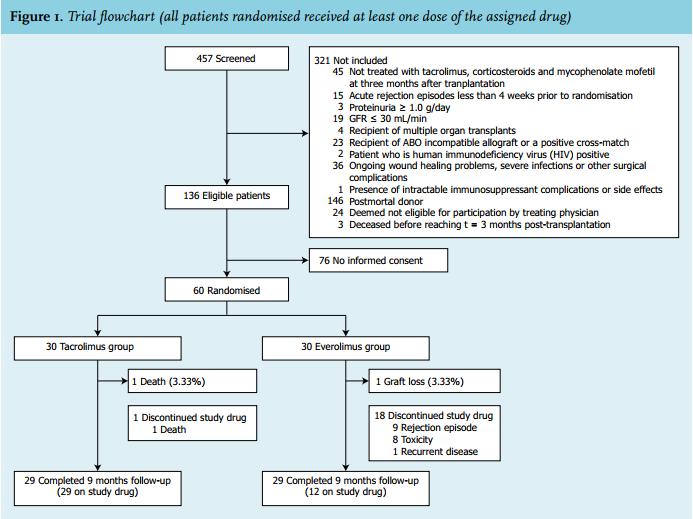
Article: Conversion from tacrolimus to everolimus with complete and early glucocorticoid withdrawal after kidney transplantation: a randomised trial (full text) - January 2018 - NJM
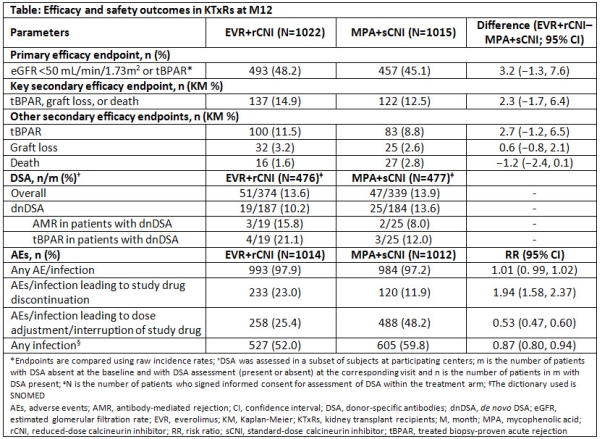
Everolimus with Reduced Calcineurin Inhibitor Exposure in De Novo Kidney Transplant Recipients: Efficacy and Safety Outcomes from the TRANSFORM Study - ATC Abstracts

Everolimus-based, calcineurin-inhibitor-free regimen in recipients of de-novo kidney transplants: an open-label, randomised, controlled trial - The Lancet
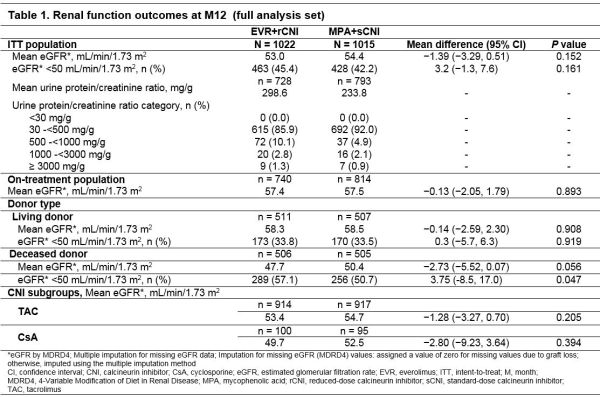
Everolimus with Reduced-Dose Calcineurin Inhibitor versus Mycophenolate with Standard-Dose Calcineurin Inhibitor in De Novo Kidney Transplant Recipients: Renal Function Results from the TRANSFORM Study - ATC Abstracts
Mechanistic analyses in kidney transplant recipients prospectively randomized to two steroid free regimen—Low dose Tacrolimus with Everolimus versus standard dose Tacrolimus with Mycophenolate Mofetil | PLOS ONE

Figure 1 from Efficacy and Safety of an Everolimus- vs. a Mycophenolate Mofetil-Based Regimen in Pediatric Renal Transplant Recipients | Semantic Scholar

De Novo Therapy with Everolimus and Low-Dose Calcineurin Inhibitors in Kidney Transplantation | Semantic Scholar
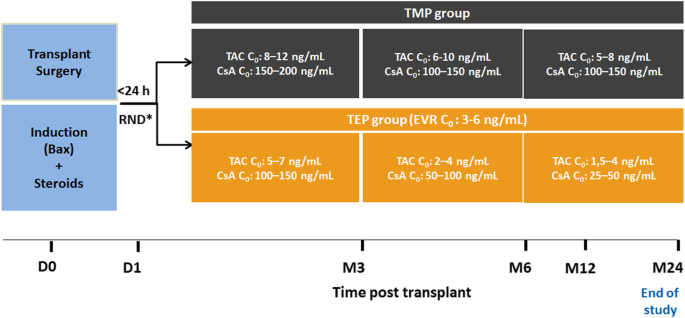
Rationale and design of the OPTIMIZE trial: OPen label multicenter randomized trial comparing standard IMmunosuppression with tacrolimus and mycophenolate mofetil with a low exposure tacrolimus regimen In combination with everolimus in de